
Rheology modification
Rheology is the study of deformation and flow of matter under applied forces. This flow and deformation can be measured using various rheological methods. Besides viscosity, the structure and solid-like properties of the material play a crucial role in product attributes and behaviours. Often the rheology of products for the household, industrial and institutional cleaning market has to be adjusted to fit the needs of each application. Therefore customers find that they often need to adjust the rheology of formulations using rheology modifiers.

Home Care product guide

Water-based and oil-based formulations
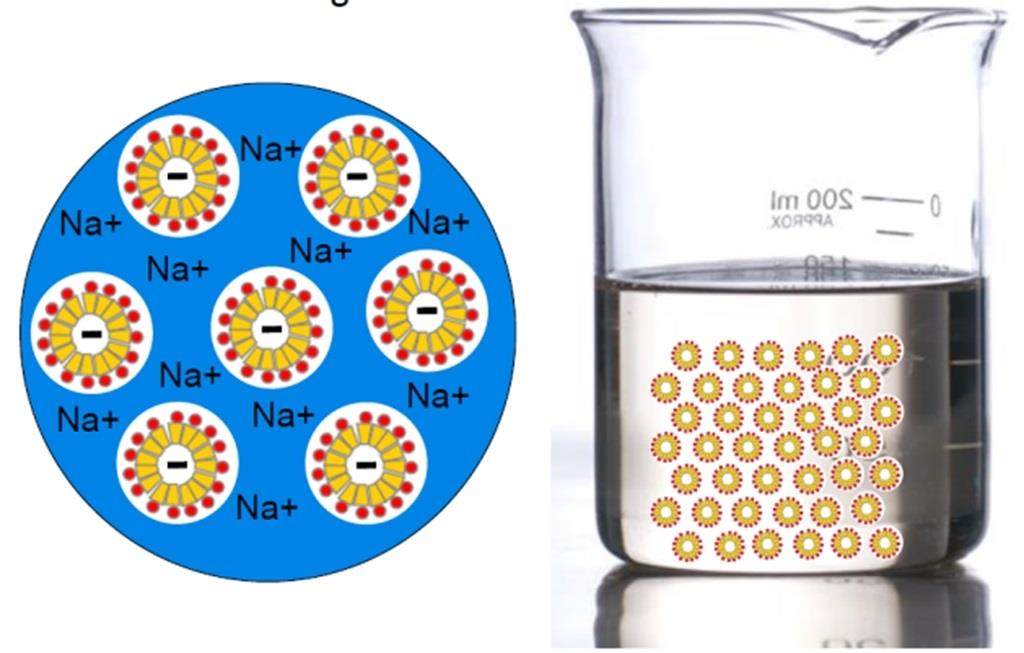
Rheology modifiers can be used for water-based formulations, as well as oil-based formulations. Water-based formulations are often thickened with standard rheology modifiers like salt or hydrophilic polymers. In oil-based formulations hydrophobic polymers, waxes or inorganic gellants are generally used. The thickening effect of salt is caused by ionic interactions between negatively charged micelles of anionic surfactants and sodium cations of the salt, shown in the image to the left. This effect is strongly dependent on the concentration of salt and anionic surfactants in the formulation.
Rheology modification with hydrophilic polymers is caused by entanglement and electrostatic counteractions which cause the polymers to build 3D-structures. The supramolecular structure influences the rheological properties of the formulation significantly. Thus it is possible to obtain formulations with high viscosity due to gel formation of the polymer. Also an increased stabilisation of particles and hydrophobic oils can be achieved.
Depending on the type of rheology modifier, when the formulation is put under high shear stress it can cause a rapid drop in viscosity - this effect is known as shear thinning and can be observed for our range polymeric rheology modifiers.

CrystaSense Sapphire 'spray and stick' technology
